Context
With a view to developing biological or medical knowledge, clinical research allows the deployment of scientific studies carried out on humans. At the same time, the management of antibiotics in intensive care and emergency medicine must take into account several constraints. In intensive care, the prescription of antibiotic therapy has a certain number of particularities. As human health is not immune to antibiotic resistance, it is important to focus on this particular area.
This is why, as part of its missions, PROMISE is carrying out an epidemiological study called “Constantine”, relating to the use of antibiotic therapy in France in several clinical research networks involved in the project.
Objectives
The “Constantine” epidemiological study involves four intensive care and infectious disease clinical research networks in France, members of PROMISE:
- OUTCOME REA (Paris), which develops research and teaching actions to improve the care of the most serious patients;
- REA-REZO (Lyon), which offers a multicenter system for monitoring and evaluating the risk of infectious diseases associated with care and antibiotic resistance, specific to adult intensive care;
- CRICS-TRIGGERSEP (franco-belgian network), which focuses on clinical research in intensive care – resuscitation and mainly focuses its research activity on sepsis;
- CIC LIMOGES (Limoges), which promotes the development of clinical research in collaboration with the themes of excellence of the University Hospital Center.
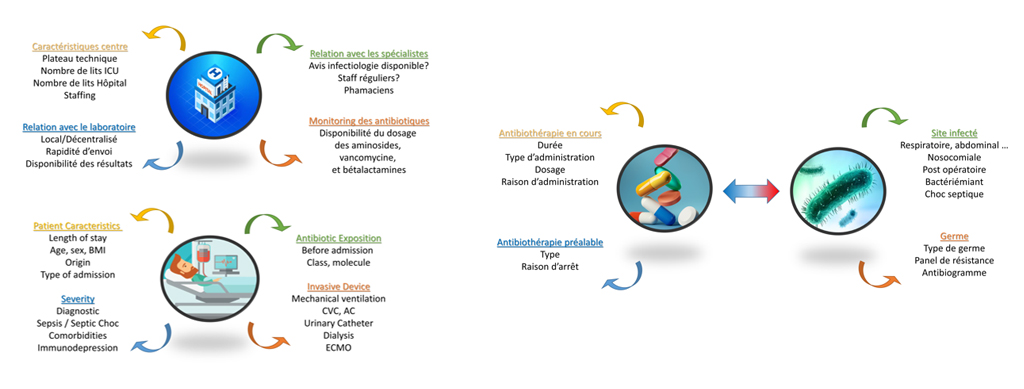
Its main objective is the description of the use of antibiotic therapy in intensive care, with a global and targeted vision of the patient. The added value of this inter-network “one-shot hospital correlation” study lies in taking into account a large patient volume, as well as follow-up after 30 days. The key figures associated with this study will be available soon.
Related media [FR]
The structuring elements of the “Constantine” epidemiological study